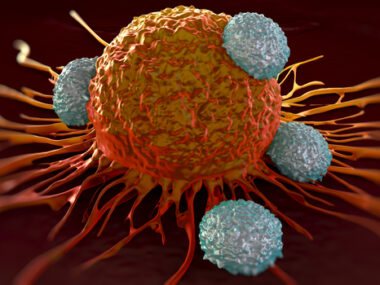
A new approach using genetically modified T-cells — via CRISPR-Cas9 gene editing technology — safely and effectively targets cancer cells in patients with advanced…
Myeloma is a rare blood cancer that begins in plasma cells, a type of white blood cell normally responsible for producing antibodies that help fight off infectious pathogens and other threats.
While myeloma treatment can drive the disease into remission, sometimes for long periods of time, the cancer will often come back after a few months or years, and additional treatments will be needed.

A new approach using genetically modified T-cells — via CRISPR-Cas9 gene editing technology — safely and effectively targets cancer cells in patients with advanced…

Nektar Therapeutics is launching a first-in-human Phase 1 clinical trial to assess the safety and efficacy of…

MYELOMA
The U.S. Food and Drug Administration (FDA) has granted orphan drug status to Teneobio‘s bispecific antibody TNB-383B as a possible treatment of multiple myeloma. Orphan…

U.S. blood cancer specialists are aware of recent approvals for multiple myeloma patients and expect to increase their use of Darzalex (daratumumab) as…

MYELOMA
Health Canada has approved Darzalex (daratumumab), in combination with standard Revlimid (lenalidomide) and dexamethasone, for people newly diagnosed with multiple myeloma who…

With the goal of encouraging the multiple myeloma community while raising money to advance treatments, Moving Mountains for Multiple Myeloma (MM4MM) is gearing up for its…
Don't have an account?
|
Already have an account?
Create your account by filling in the information below:
By creating an account, you are agreeing to the Privacy Policy and Terms of Service.