Xpovio (selinexor) for multiple myeloma
What is Xpovio for multiple myeloma?
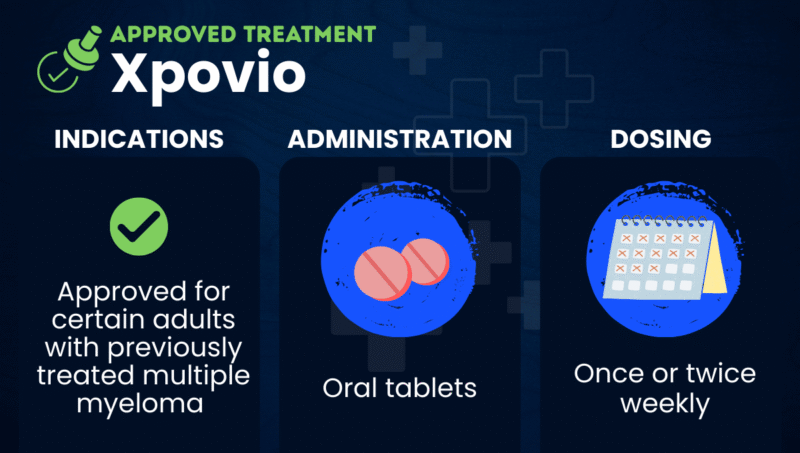
Xpovio (selinexor) is an oral targeted therapy approved to treat certain adults with multiple myeloma who have previously received other medications. It’s available as oral tablets, which are usually taken once or twice weekly.
Specifically, Xpovio is approved in combination with Velcade (bortezomib) and dexamethasone for adults who have received at least one prior therapy. It can also be used in combination with dexamethasone alone for adults who have received at least four prior therapies and whose disease is refractory, or has failed to respond, to several medications in different classes of treatment.
In multiple myeloma, immune cells called plasma cells — which normally help fight infections — become cancerous and grow uncontrollably in the bone marrow, the spongy tissue inside some bones.
The cancerous multiple myeloma cells rely on a protein called exportin 1 (XPO1) to shuttle certain regulatory proteins from the nucleus — where a cell’s genetic material is stored — into the surrounding cytoplasm. Among the proteins transported out of the nucleus are tumor-suppressor proteins, which act as natural anti-cancer proteins that trigger the death of abnormal cells.
In many myeloma cells, XPO1 is overactive, moving tumor-suppressor proteins out of the nucleus more rapidly and weakening their ability to keep cancer growth in check.
Xpovio works by blocking XPO1 activity to disrupt this protein transport. As a result, tumor-suppressor proteins remain in the nucleus, where they can exert their normal functions, including slowing cell growth and promoting cancer cell death.
Xpovio was developed and is sold in the U.S. by Karyopharm Therapeutics. In Europe, it is marketed by Stemline Therapeutics under the brand name Nexpovio.
In addition to myeloma, Xpovio is also approved for another blood cancer, diffuse large B-cell lymphoma.
Therapy snapshot
| Brand name | Xpovio |
| Chemical name | Selinexor |
| Usage | Used in combination with other therapies for adults with multiple myeloma who have received prior treatment |
| Administration | Oral tablets |
Who with multiple myeloma can take Xpovio?
Xpovio is approved in the U.S. for previously treated adults with multiple myeloma in two specific indications:
- In combination with Velcade and dexamethasone for patients who have received at least one prior therapy.
- In combination with dexamethasone for patients who have received at least four prior therapies and have not responded to at least two proteasome inhibitors, at least two immunomodulatory therapies, and one anti-CD38 antibody.
The prescribing information for Xpovio does not list any contraindications, or reasons not to use the therapy.
How is Xpovio administered in multiple myeloma?
Xpovio is available as oral tablets, which should be swallowed whole with water and not crushed, chewed, broken, or divided. Tablets are typically taken once or twice weekly, but dosing and treatment schedule depend on the indication:
- When taken with Velcade and dexamethasone, Xpovio is usually taken at 100 mg once weekly.
- When taken with dexamethasone alone, Xpovio is typically used at a recommended dose of 80 mg on days 1 and 3 each week.
Dose adjustments may be needed if patients experience certain side effects or show alarming reductions in blood cell counts.
Xpovio in multiple myeloma clinical trials
Two clinical trials mainly supported Xpovio’s approvals for multiple myeloma in the U.S.
- The BOSTON Phase 3 trial (NCT03110562) evaluated Xpovio in combination with Velcade and dexamethasone in patients who had received one to three prior therapies. This study showed that the triple combination significantly extended the time to disease progression or death compared with Velcade plus dexamethasone alone. Overall response rates were also significantly higher with Xpovio.
- The STORM Phase 2 study (NCT02336815) investigated Xpovio in combination with dexamethasone in patients with relapsed or refractory multiple myeloma who had received multiple prior regimens. Investigators measured the overall response rate in people whose disease was refractory to two proteasome inhibitors, two immunomodulatory therapies, and one anti-CD38 antibody as the primary endpoint. Results showed that 25% of patients responded at least partially to treatment.
Xpovio side effects
Xpovio’s side effect profile varies depending on the combination used. When used with Velcade and dexamethasone, common side effects include:
- fatigue
- nausea
- decreased appetite
- diarrhea
- damage to nerve cells outside of the brain and spinal cord, which may cause pain and abnormal sensations
- upper respiratory tract infections
- weight loss
- cataracts, or a clouding of the eye’s natural lens
- vomiting
- low levels of certain blood components, including platelets, red blood cells, neutrophils, and lymphocytes
- low phosphate or sodium levels
When used with dexamethasone alone, typical side effects include:
- fatigue
- nausea
- low levels of certain blood cells, including red blood cells, neutrophils, and white blood cells
- decreased appetite
- weight loss
- diarrhea
- vomiting
- low sodium levels
- constipation
- shortness of breath
- upper respiratory tract infections
The prescribing information also lists warnings for several more severe side effects, for which careful monitoring or adjustments to treatment may be needed. These include:
- serious stomach and gut problems
- serious infections, some of which may be fatal
- nervous system problems
Patients should also undergo regular monitoring to identify potentially severe changes in blood cell counts or dangerously low sodium levels, which can be life-threatening.
Xpovio may also cause harm to a developing fetus or embryo, so people who have, or whose partner has, the potential to become pregnant should use effective contraception while taking the medication and for at least one week after stopping.
Rare Cancer News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by