Velcade (bortezomib) for multiple myeloma
What is Velcade for multiple myeloma?
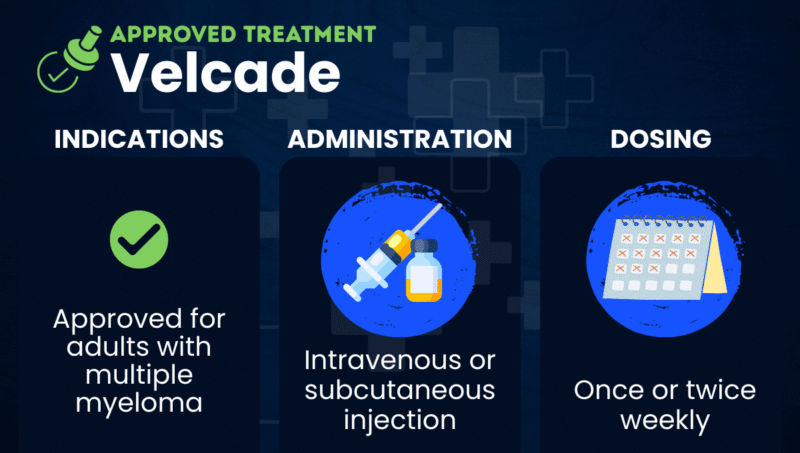
Velcade (bortezomib) is a proteasome inhibitor approved to treat adults with multiple myeloma, either alone or in combination with other treatments.
It is taken as an intravenous (into-the-vein) or subcutaneous (under-the-skin) injection, typically once or twice weekly, depending on the treatment regimen.
Velcade works by inhibiting the proteasome, a protein complex that degrades damaged or misfolded proteins within cells. By interfering with this process, the therapy causes proteins to accumulate within myeloma cells, leading to cellular stress and cell death. Because myeloma cells produce large amounts of proteins, they are more sensitive to proteasome inhibition.
Velcade is marketed in the U.S. by Takeda and in Europe and other regions by Janssen, a subsidiary of Johnson & Johnson. Generic versions are also available.
In addition to multiple myeloma, Velcade is also approved to treat adults with another type of blood cancer called mantle cell lymphoma.
Therapy snapshot
| Brand name | Velcade |
| Chemical name | Bortezomib |
| Usage | Used to treat adults with multiple myeloma |
| Administration | Subcutaneous or intravenous injection |
Who with multiple myeloma can take Velcade?
In the U.S., Velcade is approved for adults with multiple myeloma in several treatment settings.
According to its label, Velcade is approved for use in combination with the chemotherapy agent melphalan and the corticosteroid prednisone for people with newly diagnosed multiple myeloma. It may also be used as a standalone agent after at least one prior therapy for people who have previously responded to Velcade and have relapsed at least six months later.
In clinical practice, Velcade is also commonly used as part of various combination regimens for both newly diagnosed and previously treated patients. Some of these combinations are described in the prescribing information for other therapies rather than in Velcade’s own label. Examples include regimens that pair Velcade with immunomodulatory agents, monoclonal antibodies, corticosteroids, or chemotherapy drugs.
Depending on the treatment plan, Velcade may be used during different phases of care, such as initial therapy, consolidation after stem cell transplant, maintenance therapy, or in patients with relapsed or refractory disease.
In the U.S., Velcade is contraindicated for people who have experienced a hypersensitivity reaction (harmful immune responses, including allergic reactions) to bortezomib, boron, or mannitol. The therapy must not be administered intrathecally (into the spinal canal), as this can be fatal.
How is Velcade administered in multiple myeloma?
Velcade is given as either a subcutaneous or intravenous injection, which typically takes only a few seconds to administer. A healthcare professional normally gives the therapy in a clinical setting, but some patients may be able to administer subcutaneous injections at home after receiving proper training.
The recommended dose of Velcade is 1.3 mg per square meter of body surface area, though dose adjustments may be needed for some patients. Treatment schedules vary depending on the clinical setting and combination regimen.
Velcade is generally given once or twice weekly in repeating treatment cycles that may last up to six weeks. These cycles often include scheduled rest periods to allow the body to recover between treatments.
Velcade in multiple myeloma clinical trials
Several clinical trials supported the U.S. approval of Velcade, both as a single agent and in combination regimens for multiple myeloma:
- The Phase 3 VISTA trial (NCT00111319) evaluated Velcade in combination with melphalan and prednisone in previously untreated patients. Results showed that the Velcade-based regimen significantly extended the time to disease progression compared with melphalan and prednisone alone. Overall survival and overall response rates were also significantly better with Velcade.
- The Phase 3 APEX trial (NCT00048230) compared Velcade alone with the corticosteroid dexamethasone in patients whose disease had progressed after one to three prior therapies. In these patients, Velcade significantly improved the time to disease progression and response rates compared with dexamethasone.
- The MMY-3021 Phase 3 trial (NCT00722566) compared subcutaneous and intravenous administration of Velcade. The study found that both methods were equally effective, with no differences in time to progression, survival, or overall response rates.
- A Phase 2 trial (NCT00431769) assessed retreatment with Velcade in patients who had previously responded to the therapy. It showed that Velcade was effective and generally well tolerated in this setting.
Several other trials have supported Velcade’s approval in combination with other treatments.
Velcade side effects
The most common side effects of Velcade include:
- nausea
- diarrhea
- low levels of certain blood components, including platelets, red blood cells, neutrophils, lymphocytes, and white blood cells
- damage to nerve cells outside of the brain and spinal cord, which may cause pain and abnormal sensations
- fatigue
- constipation
- vomiting
- rash
- fever
- loss of appetite
Some people taking Velcade may experience more serious or potentially life-threatening side effects, for which careful monitoring or adjustments to their treatment may be necessary:
- low blood pressure
- severe heart, lung, gastrointestinal tract, or liver complications, including organ failure
- neurological symptoms and altered mental status due to swelling in the brain
- tumor lysis syndrome, a serious condition that can occur when many cancer cells die rapidly
- thrombotic microangiopathy, a condition marked by blood clots in small blood vessels that may lead to organ damage
Velcade may also harm a developing fetus, so effective contraception is recommended during treatment, and for several months after stopping, if a pregnancy is possible for the patient or their partner.
Rare Cancer News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by