Thalomid (thalidomide) for multiple myeloma
What is Thalomid for multiple myeloma?
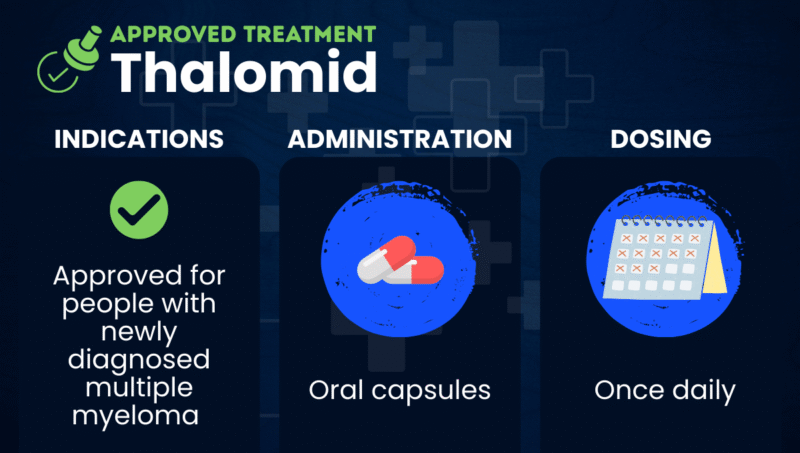
Thalomid (thalidomide) is an oral immunomodulatory treatment that’s approved for people with newly diagnosed multiple myeloma when used in combination with the corticosteroid dexamethasone.
Multiple myeloma is a type of cancer that develops when plasma cells — immune cells that normally help fight infections — become cancerous and start growing out of control in the bone marrow, the spongy tissue inside certain bones where blood cells are made.
Thalomid works in several ways to slow the growth of cancerous plasma cells, also called myeloma cells. For example, it can reduce the production of signaling molecules that support myeloma cells and boost the activity of certain immune cells, including T-cells and natural killer cells, which can target and destroy myeloma cells.
The therapy also blocks the growth of new blood vessels in the bone marrow, starving myeloma cells of the oxygen and nutrients they need to survive and grow.
Thalomid is marketed by Bristol Myers Squibb and is also approved for erythema nodosum leprosum, a complication of leprosy. Generics of this medication are available.
Therapy snapshot
| Brand name | Thalomid |
| Chemical name | Thalidomide |
| Usage | Used in combination with dexamethasone in people with newly diagnosed multiple myeloma |
| Administration | Oral capsules |
Who with multiple myeloma can take Thalomid?
Thalomid is approved in the U.S. for people with newly diagnosed multiple myeloma, when used in combination with dexamethasone.
However, the medication’s prescribing information carries a boxed warning noting that it can cause severe birth defects and embryonic death when taken during pregnancy and also increases the risk of serious clotting complications.
To ensure Thalomid is never taken during pregnancy, the medication is available only through a restricted program that ensures healthcare professionals and pharmacies are certified to prescribe and dispense the therapy. Patients also must sign an agreement form and comply with contraception requirements.
In the U.S., the therapy is contraindicated for patients who:
- are pregnant
- have shown hypersensitivity reactions (an exaggerated immune response) to any ingredient in Thalomid
How is Thalomid administered in multiple myeloma?
Thalomid is available as oral capsules that patients can take once daily at home. The capsules should be swallowed whole with water — and not opened or crushed — ideally at bedtime and at least one hour after a meal.
Thalomid’s recommended dose is 200 mg per day, taken in 28-day cycles. Dexamethasone is administered at a 40 mg dose on only 12 of the 28 treatment days.
Thalomid in multiple myeloma clinical trials
The U.S. approval of Thalomid in combination with dexamethasone for people newly diagnosed with multiple myeloma was supported by data from two Phase 3 clinical trials. Both compared the combination against dexamethasone plus a placebo, but dosing details and outcome measures differed.
- Study 1 tested the now-recommended dosing of Thalomid and dexamethasone, and each group received four treatment cycles. Results showed that overall response rates were significantly higher in the Thalomid group (52% vs. 36% in the placebo group), meeting the trial’s main goal.
- Study 2 (NCT00057564) started with a lower dose of Thalomid and gradually increased it to 200 mg over the first cycle. Data showed that patients who received Thalomid plus dexamethasone experienced significantly longer time to disease progression than those given a placebo plus dexamethasone (97.7 vs. 28.3 weeks). This represented a 57% reduction in the risk of progression, again meeting the trial’s main goal. The Thalomid group also had a significantly higher overall response rate (63% vs. 46%).
Thalomid side effects
The most common side effects of Thalomid reported in multiple myeloma clinical trials include:
- fatigue
- low calcium levels
- swelling
- constipation
- symptoms of nerve damage, such as tingling, prickling, or nerve pain
- shortness of breath
- muscle weakness
- low levels of certain blood cells, including white blood cells and neutrophils
- rash or peeling skin
- confusion
- loss of appetite
- nausea
- anxiety or agitation
- general weakness or reduced energy
- tremor
- fever
- weight loss or weight gain
- blood clots
- dizziness
- dry skin
Thalomid also carries a boxed warning noting it can cause serious issues, including:
- severe birth defects and death of embryos or fetuses if taken during pregnancy. Females of reproductive potential must undergo routine pregnancy tests and avoid pregnancy during treatment. They must also abstain from sexual intercourse or use two reliable methods of contraception to prevent pregnancy during this period. Because Thalomid can be found in semen, males must also use an effective method of contraception.
- serious blood clotting issues, including deep vein thrombosis and pulmonary embolism, in which a blood clot forms in a deep vein or in the lungs, respectively. In one clinical trial, nearly one quarter of patients taking a combination of Thalomid and dexamethasone experienced these clotting issues. Depending on a person’s risk factors, doctors may recommend clot-preventive therapies to reduce this risk.
Other serious side effects that may occur while on treatment with Thalomid include:
- cardiovascular problems, including stroke and heart disease
- severe drowsiness
- damage to nerves outside the brain and spinal cord
- dizziness or a drop in blood pressure upon standing
- severe reductions in levels of neutrophils or platelets, cell fragments that help the blood to clot
- an increase in viral load for people with HIV infection
- slowed heart rate
- severe skin reactions
- seizures
- hypersensitivity reactions, including swelling and a severe reaction called anaphylaxis
- tumor lysis syndrome, a serious condition that occurs when cancer cells die too rapidly
Finally, adding the cancer antibody therapy Keytruda (pembrolizumab) to dexamethasone and Thalomid may increase the risk of death.
Rare Cancer News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by