Talvey (talquetamab-tgvs) for multiple myeloma
What is Talvey for multiple myeloma?
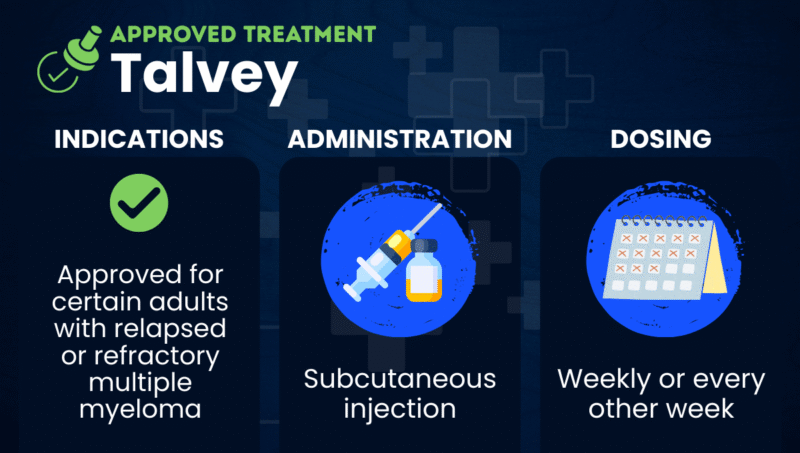
Talvey (talquetamab-tgvs) is an antibody-based therapy approved to treat certain adults with relapsed or refractory multiple myeloma, meaning their cancer has either returned after a period of improvement or has not responded to treatment.
Specifically, Talvey has received accelerated approval in the U.S. for patients ages 18 and older who have received at least four prior lines of therapy, based on response rates and duration of responses. Continued approval for this indication may be dependent on additional trials confirming the drug’s benefit.
In multiple myeloma, immune cells called plasma cells, which normally help the body fight infections, become cancerous and grow out of control. While the immune system has the ability to kill these cells when they become abnormal, immune cells can’t always recognize multiple myeloma cells on their own.
Talvey is a bispecific antibody designed to help immune T-cells recognize and destroy cancerous cells. It does so by simultaneously targeting the CD3 protein on the surface of T-cells and GPRC5D on plasma cells, acting as a bridge connecting both cell types. This activates T-cells and triggers the release of toxic proteins that destroy myeloma cells.
Talvey is designed to be given once or twice weekly via subcutaneous (under-the-skin) injections after an initial step-up dosing period. It was developed by Johnson & Johnson, which is now sponsoring clinical trials that could support full approval of the medication.
Therapy snapshot
| Brand name | Talvey |
| Chemical name | Talquetamab-tgvs |
| Usage | Used for certain adults with relapsed or refractory multiple myeloma |
| Administration | Subcutaneous injection |
Who can take Talvey?
Under the accelerated approval program, Talvey is approved in the U.S. for treating adults with relapsed or refractory multiple myeloma who have previously received at least four lines of therapy, including a proteasome inhibitor, an immunomodulatory drug, and an anti-CD38 antibody.
The therapy is also conditionally approved in Europe, where it is used for people with relapsed or refractory disease who have received at least three previous therapies.
There are no contraindications to using Talvey, but the medication’s prescribing information carries a boxed warning noting it can cause potentially severe or life-threatening side effects, including serious immune reactions and neurological complications.
Because of these risks, Talvey is available only through a restricted access program that ensures healthcare professionals and pharmacies are certified to prescribe and dispense the therapy.
How is Talvey administered?
Talvey is given as a subcutaneous injection, usually in the abdominal area. Injections should be administered only by a healthcare professional experienced in managing severe immune-related reactions.
A patient’s healthcare provider will decide whether treatment should be given as a 0.4 mg/kg weekly injection or as a 0.8 mg/kg every-two-weeks injection.
Regardless of treatment schedule, all patients begin with a step-up dosing period, during which doses are gradually increased over 7-10 days. This is done to reduce the risk and severity of cytokine release syndrome (CRS) — a potentially life-threatening immune reaction that sometimes occurs with certain immunotherapies.
Premedications, such as corticosteroids, antihistamines, and antifever medications, are given before step-up doses to help reduce the risk of CRS. After each step-up dose, patients must remain in a healthcare facility for 48 hours of monitoring for signs and symptoms of CRS or neurologic toxicity.
Once the step-up period is completed and the full treatment dose is reached, Talvey continues to be administered by a healthcare provider until disease progression or unacceptable toxicity.
Talvey in clinical trials
Talvey’s approval in the U.S. was mainly supported by data from the open-label Phase 1/2 MonumenTAL-1 studies (NCT03399799 and NCT04634552), which enrolled heavily pretreated adults with relapsed or refractory multiple myeloma. Eligible participants had received at least three prior therapies, including a proteasome inhibitor, an immunomodulatory agent, and an anti-CD38 antibody.
- In the primary analysis, patients given Talvey at the approved doses achieved high rates of tumor response. Among people who had not previously received T-cell redirection therapies, the overall response rate was 73% with the once-weekly (0.4 mg/kg) schedule and 73.6% with the every-other-week (0.8 mg/kg) schedule. Median duration of response was about 9.5 months with the weekly regimen. With the every-other-week regimen, an estimated 85% of responders maintained their response for at least nine months.
- Among those previously exposed to T-cell redirection therapies given the weekly schedule, the overall response rate was 72%. An estimated 59% of responders maintained their response for nine months.
- More recent, longer-term follow-up analyses have shown that responses are durable, including in patients who received prior T-cell redirection therapy. In these analyses, the median duration of response was 17.9 months, or nearly 1,5 years, for patients not exposed to these therapies and 19.2 months, or slightly longer, for those exposed.
Several clinical studies are now ongoing to evaluate Talvey in various combination regimens, and support its full approval for multiple myeloma.
Talvey side effects
The most common side effects of Talvey include:
- fever
- CRS
- a metallic or bitter taste in the mouth
- changes in the color, shape, or texture of fingernails or toenails
- musculoskeletal pain
- skin disorders
- rash
- fatigue
- weight loss
- dry mouth
- dry skin
- difficulty swallowing
- upper respiratory tract infection
- diarrhea
- low blood pressure
- headache
- low blood cell counts
Talvey also carries a boxed warning noting it can cause serious issues due to the excess activation of the immune system, including:
- CRS, including life-threatening or fatal reactions: Patients should follow the appropriate step-up dosing to reduce the risk of CRS and pause or stop Talvey if signs of severe CRS appear.
- Neurologic toxicity, including immune effector cell-associated neurotoxicity syndrome (ICANS): Patients should be monitored for signs of neurological toxicity during treatment. If any of those symptoms emerge, Talvey should be paused or discontinued depending on severity.
Other serious side effects that may occur while on treatment with Talvey include:
- infections
- oral and skin toxicity
- signs of liver damage
The medication may also harm a developing fetus, so women with the potential to become pregnant should use effective contraception throughout treatment and for three months after stopping.
Rare Cancer News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by