Revlimid (lenalidomide) for multiple myeloma
What is Revlimid for multiple myeloma?
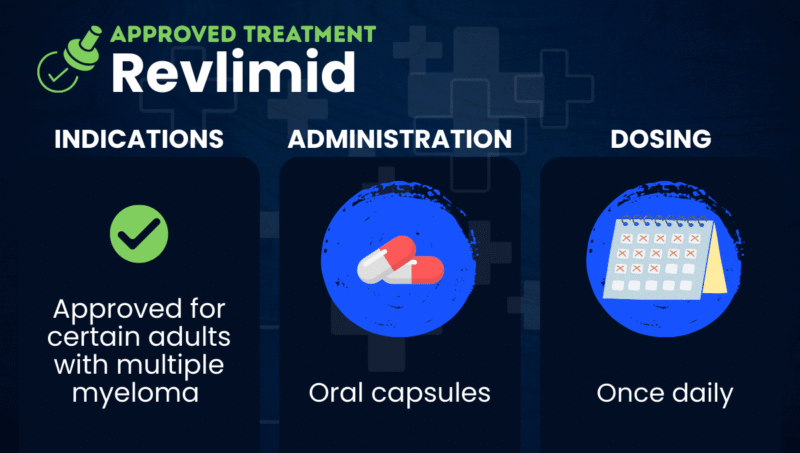
Revlimid (lenalidomide) is an oral immunomodulatory treatment that’s approved for certain adults with multiple myeloma when used in combination with the corticosteroid dexamethasone, or as a maintenance therapy following an autologous hematopoietic stem cell transplant.
Multiple myeloma is a type of cancer in which plasma cells, a type of immune cells, become cancerous and start growing out of control in the bone marrow, the spongy tissue inside some bones where blood cells are produced.
Revlimid acts in different ways to slow down the growth of cancerous plasma cells (myeloma cells). It degrades proteins that promote myeloma cell survival and suppresses proinflammatory and promyeloma signaling molecules. It also increases the activity of certain immune cells, including T-cells and natural killer cells, which in turn can attack and kill myeloma cells. Additionally, the medication can affect the environment within the bone marrow, cutting off the blood supply that helps myeloma cells grow.
Bristol Myers Squibb began marketing Revlimid after acquiring its original developer, Celgene. Generics of the medication are also available.
In addition to multiple myeloma, Revlimid is approved to treat adults with other types of blood cancer, including several types of lymphoma.
Therapy snapshot
| Brand name | Revlimid |
| Chemical name | Lenalidomide |
| Usage | Used to treat certain adults with multiple myeloma |
| Administration | Oral capsules |
Who with multiple myeloma can take Revlimid?
In the U.S., Revlimid is approved to treat adults with multiple myeloma when used:
- in combination with dexamethasone for a broad population of patients, including those who have previously received at least one therapy and those who were newly diagnosed but not eligible to receive a stem cell transplant
- as a maintenance therapy for patients who have previously received a stem cell transplant
Revlimid is contraindicated, or shouldn’t be used, by pregnant women and people who have previously had severe immune reactions to the therapy or its key ingredient.
Revlimid comes with a boxed warning that it may cause birth defects or fetal death. For this reason, it is only available in the U.S. through a restricted distribution program called Lenalidomide Risk Evaluation and Mitigation Strategy (REMS).
The boxed warning also notes that Revlimid can cause significant neutropenia, or low neutrophil counts, as well as thrombocytopenia, which is marked by low platelet counts. Neutrophils are a type of infection-fighting immune cell. Platelets are small cell fragments involved in blood clotting.
Patients taking Revlimid are also at high risk of experiencing several blood-clotting events, so doctors may recommend clot-preventive therapies to reduce this risk.
How is Revlimid administered in multiple myeloma?
Revlimid comes in the form of oral capsules that patients can take at home, with or without food. The capsules should be swallowed whole with water, and not opened, broken, or chewed. The precise dosing regimen depends on the usage:
- When used in combination with dexamethasone, the recommended starting dose of Revlimid is 25 mg daily for 21 days out of a 28-day cycle. Treatment should generally continue until disease progression or unacceptable toxicity. If patients are eligible for a stem cell transplant, stem cell mobilization should occur within the first four treatment cycles.
- When used as a maintenance therapy after a stem cell transplant, the recommended starting dose of Revlimid is 10 mg daily continuously for the whole 28 days of a cycle. After three cycles, the dose may be increased to 15 mg daily if patients tolerate it.
Dose adjustments may be required in patients with kidney function impairments, as well as in those with neutropenia or thrombocytopenia.
In female patients, pregnancy must be excluded with two negative pregnancy tests before treatment is started.
Revlimid in multiple myeloma clinical trials
Several clinical trials supported approval of Revlimid in the U.S. for various indications in multiple myeloma:
- The Phase 3 FIRST trial (NCT00689936) showed that, in newly diagnosed multiple myeloma patients who were not eligible for a stem cell transplant, a combination of Revlimid plus dexamethasone significantly reduced the risk of disease progression or death compared with a standard regimen. Data also showed that repeating treatment cycles until disease progression led to better outcomes than using a fixed number of treatment cycles.
- Two Phase 3 trials, one in North America (NCT00056160) and the other outside North America (NCT00424047), assessed the safety and effectiveness of a combination of Revlimid and dexamethasone in multiple myeloma patients who had already received at least one therapy. Both found that the time to disease progression was significantly longer in participants treated with Revlimid plus dexamethasone versus those treated with dexamethasone alone. Overall response rates were also significantly higher in the Revlimid combination groups.
- Another set of two Phase 3 trials, one based in the U.S. (NCT00114101) and the other in Europe (NCT00430365) examined Revlimid as a maintenance therapy for people who had already received induction therapy and a stem cell transplant. Both found that Revlimid maintenance therapy significantly reduced the risk of disease progression or death.
Other trials have supported Revlimid’s approval as part of combination treatments.
Revlimid side effects
The most common side effects of Revlimid in people with multiple myeloma include:
- diarrhea
- fatigue
- constipation
- low blood cell counts
- swelling in the limbs
- insomnia
- muscle cramps or spasms
- abdominal pain
- back pain
- nausea
- weakness
- fever
- upper respiratory tract infections
- bronchitis
- the common cold
- inflammation of the gastrointestinal tract
- cough
- rash
- shortness of breath
- decreased appetite
- tremor
- dizziness
Revlimid’s prescribing information also carries warnings for potentially serious side effects, including:
- increased risk of developing another type of cancer
- liver toxicity
- severe skin reactions
- tumor lysis syndrome, a serious condition that occurs when many cancer cells die rapidly, and is more likely to happen in people with a high disease burden
- low numbers of blood cell precursors, which can compromise a stem cell transplant
- allergic reactions, including swelling and a severe reaction called anaphylaxis
If a patient experiences severe skin reactions or allergic reactions, Revlimid should be permanently discontinued. Patients should also be monitored for signs of liver toxicity or changes in blood cell counts, which may require treatment interruption or dose adjustments.
People with multiple myeloma are at an increased risk of experiencing complications associated with blood clotting, such as deep vein thrombosis and stroke, when taking Revlimid alongside dexamethasone. Doctors may recommend therapies to prevent the formation of dangerous blood clots while patients are receiving treatment to reduce the risk of blood clot-related events. Patients should also be closely monitored for signs and symptoms of such events and seek immediate medical care if they experience shortness of breath, chest pain, or swelling in the limbs.
Pregnant women should not take Revlimid, as the therapy may cause birth defects or fetal death if taken during pregnancy. Females of reproductive potential must avoid pregnancy during treatment and for at least four weeks before and after finishing treatment. They must also abstain from sexual intercourse or use two reliable methods of contraception to prevent pregnancy during this period.
Because Revlimid can also be found in semen, males of reproductive potential taking the medication must also use an effective method of contraception when having sexual intercourse with a female of reproductive potential while taking the medication and for at least four weeks after stopping treatment. Men must also not donate sperm during this period.
Patients must also abstain from donating blood during treatment and for at least four weeks after finishing treatment to avoid the possibility of the blood being given to a pregnant woman whose baby would then be exposed to the medication.
Adding the cancer antibody therapy Keytruda (pembrolizumab) to dexamethasone and medications like Revlimid, which are structurally similar to Thalomid (thalidomide), may increase the risk of mortality.
Rare Cancer News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by