Ninlaro (ixazomib) for multiple myeloma
What is Ninlaro for multiple myeloma?
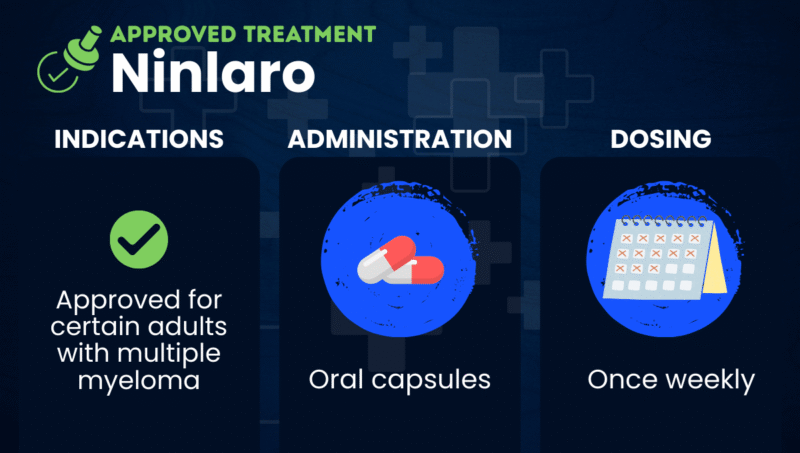
Ninlaro (ixazomib) is an oral proteasome inhibitor that’s approved to treat adults with multiple myeloma who have received at least one prior therapy. It is given in combination with Revlimid (lenalidomide) and the corticosteroid dexamethasone.
Multiple myeloma is a blood cancer that affects plasma cells, a type of white blood cell found in the bone marrow.
Ninlaro works by blocking the action of proteasomes — protein complexes found inside cells that normally break down proteins that are faulty or no longer needed. By inhibiting proteasome activity, Ninlaro causes proteins to accumulate in cancerous plasma cells, leading to cellular stress and death. This helps slow the growth and spread of myeloma cells.
Marketed by Takeda Pharmaceuticals, Ninlaro was the first oral proteasome inhibitor to be approved for multiple myeloma.
Therapy snapshot
| Brand name | Ninlaro |
| Chemical name | Ixazomib |
| Usage | Used to treat certain adults with multiple myeloma |
| Administration | Oral capsules |
Who can take Ninlaro?
In the U.S., Ninlaro is approved to be used in combination with Revlimid and dexamethasone to treat adults with multiple myeloma who have received at least one prior therapy.
Ninlaro’s U.S. prescribing information lists no contraindications to its use. However, the therapy is not indicated for maintenance treatment or for use in newly diagnosed patients.
How is Ninlaro administered?
Ninlaro is available as oral capsules that should be swallowed whole with water and not crushed, chewed, or opened. Capsules should be taken on an empty stomach, at least one hour before or two hours after meals.
The recommended starting dose of Ninlaro is 4 mg, taken once weekly on the same day each week for the first three weeks (21 days) of a four-week (28-day) treatment cycle. Treatment typically continues until disease progression or unacceptable toxicity.
Dose adjustments may be required in patients who experience certain side effects or in those who have liver or kidney function impairments.
Before starting a new treatment cycle, white blood cell and platelet levels should be high enough, and possible side effects should have mostly eased, as determined by a healthcare provider.
Ninlaro in clinical trials
Ninlaro’s approval for multiple myeloma was supported by data from the Phase 3 TOURMALINE-MM1 trial (NCT01564537), which enrolled patients whose disease had returned after treatment (relapsed) and/or had not responded adequately to previous treatment (refractory). All had received at least one prior line of therapy. Data showed that:
- adding Ninlaro to Revlimid and dexamethasone significantly reduced the risk of disease progression or death compared with Revlimid and dexamethasone alone
- after about seven years of follow-up, overall survival was similar in the two groups, but some higher-risk patients still appeared to benefit more from the Ninlaro-based treatment
Other clinical trials have also studied Ninlaro in settings beyond those covered in the label:
- The Phase 3 TOURMALINE-MM2 trial (NCT01850524) showed that patients with newly diagnosed multiple myeloma who were not eligible for a stem cell transplant lived longer without their disease worsening when treated with Ninlaro plus Revlimid and dexamethasone compared with a placebo plus Revlimid and dexamethasone. While the difference was not statistically significant, patients with high-risk disease appeared to benefit more.
- The Phase 3 TOURMALINE-MM3 trial (NCT02181413) showed that patients with newly diagnosed multiple myeloma who received Ninlaro as maintenance therapy after a stem cell transplant lived longer without their disease worsening than those who received a placebo.
- The Phase 3 TOURMALINE-MM4 trial (NCT02312258) evaluated Ninlaro as a maintenance therapy in patients with newly diagnosed multiple myeloma who did not undergo a stem cell transplant. Patients who received Ninlaro lived longer without their disease worsening compared with those who received a placebo.
Ninlaro side effects
The most common side effects of Ninlaro include:
- low levels of platelets, the small cell fragments involved in blood clotting
- low levels of neutrophils, a type of infection-fighting immune cell
- diarrhea
- constipation
- nausea or vomiting
- swelling in the hands, ankles, or feet
- nerve problems, such as numbness, tingling, or burning sensations
- bronchitis (lung irritation)
- rash
The therapy also comes with warnings that its use could cause other potentially serious adverse events, such as:
- liver toxicity
- thrombotic microangiopathy, a rare but serious disorder that causes blood clots to form in small blood vessels and can damage organs such as the kidneys and brain
- Stevens-Johnson syndrome and toxic epidermal necrolysis, rare, life-threatening skin reactions involving severe blistering, skin detachment, and mucus membrane damage
Patients should be monitored for these side effects and complications during treatment. If serious side effects occur, treatment may be delayed, the dose adjusted, or the medication discontinued.
Ninlaro can also cause fetal harm and should not be used during pregnancy. As such, patients of reproductive potential should use effective methods of contraception to avoid pregnancy during treatment and for three months after the last dose. Female patients are also advised not to breastfeed during this period.
Rare Cancer News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by