Kyprolis (carfilzomib) for multiple myeloma
What is Kyprolis for multiple myeloma?
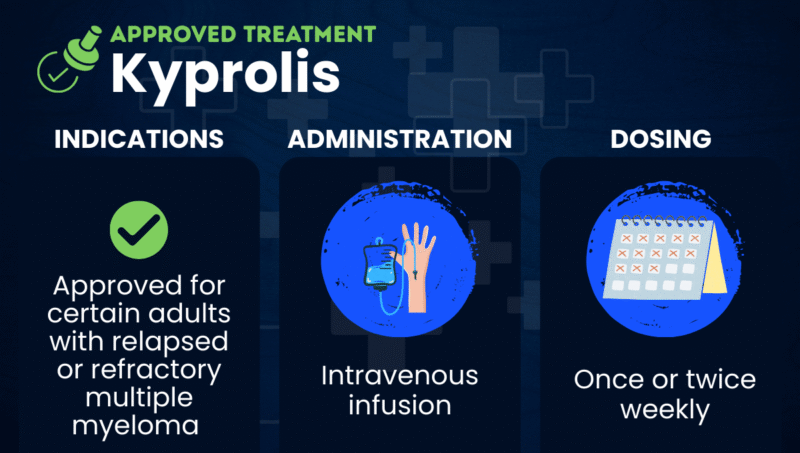
Kyprolis (carfilzomib) is a proteasome inhibitor that’s approved for certain adults with relapsed or refractory multiple myeloma (RRMM), whose disease has returned after initial treatment or has not responded to it.
The therapy can be used alone or in combination with other treatments. It is given via an intravenous (into-the-vein) infusion once or twice weekly.
Kyprolis is designed to kill cancerous myeloma cells by targeting the proteasome, a protein complex that serves as the cell’s garbage disposal system, breaking down damaged or unnecessary proteins into smaller pieces.
By blocking proteasome activity, the therapy causes proteins to accumulate inside myeloma cells, leading to cellular stress and ultimately cell death. Because myeloma cells produce large amounts of abnormal proteins, they are particularly sensitive to proteasome inhibition.
The name-brand therapy Kyprolis is marketed by Amgen, and generic versions are available.
Therapy snapshot
| Brand name | Kyprolis |
| Chemical name | Carfilzomib |
| Usage | Used alone or in combination with other therapies to treat certain adults with relapsed or refractory multiple myeloma |
| Administration | Intravenous infusion |
Who can take Kyprolis?
Kyprolis is approved in the U.S. for adults with RRMS who have received one to three prior lines of treatment in combination with other therapies, including:
- dexamethasone
- Revlimid (lenalidomide) and dexamethasone
- Darzalex (daratumumab) and dexamethasone
- Darzalex Faspro (daratumumab and hyaluronidase-fihj) and dexamethasone
- Sarclisa (isatuximab-irfc) and dexamethasone
Kyprolis is also approved as a standalone therapy for adults with RRMS who have received one or more prior lines of therapy.
The prescribing information for Kyprolis does not list any contraindications or reasons not to use the therapy.
How is Kyprolis administered?
Kyprolis is administered by a healthcare professional via an intravenous infusion lasting about 10-30 minutes, depending on the combination used.
The precise dosage and infusion schedule also depend on the treatment regimen, but Kyprolis is generally administered once or twice weekly for three weeks, followed by one week off, in repeating 28-day cycles.
Patients will usually receive a lower starting dose during the first week of their first treatment cycle. If the therapy is well tolerated, the dose is increased in the second week and maintained in subsequent weeks and cycles.
Dose adjustments or treatment interruptions may be needed if certain side effects occur. Modifications may also be required for people with liver problems.
To help reduce the risk of complications, patients may receive oral and intravenous fluids before and after Kyprolis, especially during the first cycle. Doctors typically give dexamethasone before infusions to lower the risk of infusion-related reactions. Preventive medications may be recommended to reduce the risk of blood clots and herpes zoster (shingles) reactivation.
Kyprolis in clinical trials
Several clinical trials supported the U.S. approval of Kyprolis, both as a single agent and in various combination regimens, for the treatment of relapsed or refractory multiple myeloma:
- The Phase 3 ENDEAVOR trial (NCT01568866) showed that Kyprolis plus dexamethasone significantly extended the time to disease progression compared with the standard combination of Velcade (bortezomib) and dexamethasone. Long-term analyses also showed improved overall survival with the Kyprolis regimen.
- The Phase 3 ASPIRE trial (NCT01080391) evaluated Kyprolis in combination with Revlimid and dexamethasone. Results showed the Kyprolis combination significantly extended the time patients lived without disease progression compared with Revlimid plus dexamethasone alone. Overall response rates and survival rates were also significantly higher in the Kyprolis group.
- Two studies supported approval of the combination of Kyprolis, Darzalex, and dexamethasone: the Phase 1 EQUULEUS trial (NCT01998971) and the Phase 3 CANDOR trial (NCT03158688). In EQUULEUS, all participants received the triplet regimen, and 84% experienced at least a partial response. In CANDOR, the triplet combination significantly improved progression-free survival (time to disease progression or death) compared with Kyprolis plus dexamethasone alone.
- The Phase 2 PLEIADES trial (NCT03412565) evaluated the combination of Kyprolis, Darzalex Faspro, and dexamethasone, showing an overall response rate of nearly 85%.
- The Phase 3 IKEMA trial (NCT03275285) showed that the combination of Kyprolis, Sarclisa, and dexamethasone significantly extended the time to disease progression compared with Kyprolis plus dexamethasone alone. Follow-up analyses showed a median progression-free survival of about three years with the triplet regimen, compared with less than two years with Kyprolis plus dexamethasone.
- Three single-arm trials — the Phase 1/2 PX-171-007 study (NCT00531284) and the Phase 2 studies PX-171-003 A1 (NCT00511238) and PX-171-004 Part 2 (NCT00530816) — evaluated Kyprolis as a standalone therapy. These studies demonstrated overall response rates of 23% to 50%, supporting its approval as a single agent.
Kyprolis side effects
The most common side effects reported in clinical trials testing Kyprolis as a single agent include:
- low levels of red blood cells and platelets
- fatigue
- nausea
- fever
- shortness of breath
- diarrhea
- headache
- cough
- swelling in the arms or legs
When used in combination regimens, the most common side effects of Kyprolis include:
- low levels of red blood cells and platelets
- diarrhea
- high blood pressure
- fatigue
- upper respiratory tract infection
- fever
- cough
- shortness of breath
- insomnia
The prescribing information for Kyprolis also carries several warnings for potentially serious side effects, including:
- severe heart, kidney, lung, and liver complications, including organ failure
- tumor lysis syndrome, a serious condition that can occur when many cancer cells die rapidly
- high blood pressure in the lungs
- serious blood-clotting issues
- infusion-related reactions
- severe bleeding, which can lead to death
- neurological symptoms and altered mental status due to swelling in the brain
- progressive multifocal leukoencephalopathy, a rare and potentially life-threatening brain infection
The medication may also harm a developing fetus, so effective contraception is recommended throughout treatment if pregnancy is a possibility for a patient or their partner.
Rare Cancer News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by