Iberdomide for multiple myeloma
What is iberdomide for multiple myeloma?
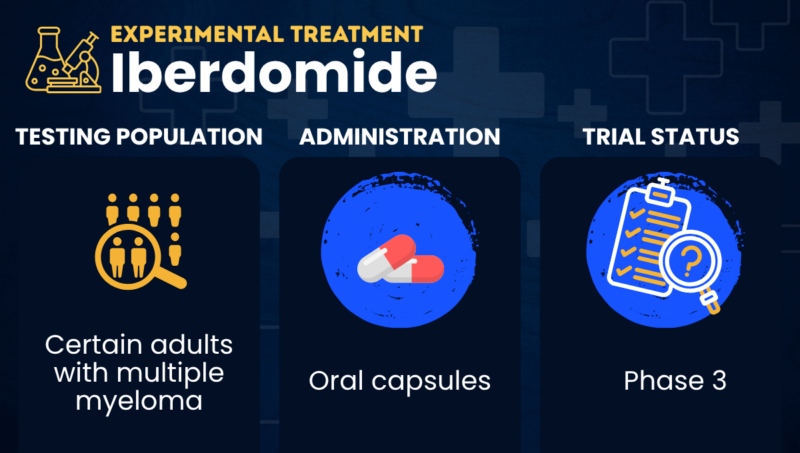
Iberdomide is an experimental oral therapy being developed by Bristol Myers Squibb for multiple myeloma.
It is under review by the U.S. Food and Drug Administration (FDA) as part of a combination regimen for certain people with myeloma who have relapsed or failed to respond to prior therapies (relapsed/refractory, or RRMM), with a decision expected in August 2026.
Multiple myeloma occurs when plasma cells, a type of immune cell, grow out of control in bone marrow, the spongy tissue inside some bones.
Iberdomide belongs to a class of next-generation immunomodulatory drugs called cereblon E3 ligase modulatory drugs, or CELMoDs. It works by binding to the protein cereblon, which is involved in the system that marks defective and abnormal proteins for recycling.
This binding enables more efficient tagging of proteins that support myeloma cell survival, expediting their degradation. Without these necessary proteins, myeloma cells die. The medication also stimulates the immune system to further attack cancerous cells.
While the regulatory application currently under review with the FDA is specifically for RRMM, Bristol Myers Squibb is also sponsoring clinical trials of the medication in other myeloma contexts, including in people who have recently received a stem cell transplant.
Therapy snapshot
| Treatment name | Iberdomide |
| Administration | Oral capsule |
| Clinical testing | Currently in Phase 3 testing |
How will iberdomide be administered in multiple myeloma?
Clinical trials have tested iberdomide as oral capsules. In an ongoing Phase 3 study, the medication is taken daily for the first three weeks of each four-week treatment cycle. While multiple doses have been tested, the optimal dose selected for further study is 1 mg.
Iberdomide in multiple myeloma clinical trials
The application under review for RRMM covers the use of iberdomide in combination with dexamethasone and daratumumab (sold under the brand names Darzalex and Darzalex Faspro in different formulations). Data from two trials support that regimen:
- The ongoing Phase 3 EXCALIBER-RRMM trial (NCT04975997) enrolled 864 people with RRMM who had previously received one or two lines of therapy. They are receiving dexamethasone and daratumumab, in combination with either iberdomide or bortezomib (sold as Velcade and generics). Early results showed that more people in the iberdomide group achieved minimal residual disease negativity, a state where no detectable myeloma cells remain.
- The earlier Phase 1/2 CC-220-MM-001 trial (NCT02773030) assessed iberdomide as part of several treatment combinations, overall involving 466 people with myeloma. Data from RRMM patients showed that iberdomide and dexamethasone demonstrated meaningful clinical activity in heavily pretreated patients, including when combined with other agents such as daratumumab. In a subset of Phase 1/2 trial participants with newly diagnosed myeloma who were not eligible for a stem cell transplant, the iberdomide, dexamethasone, and daratumumab combination also showed signs of efficacy.
The Phase 3 EXCALIBER-Maintenance trial (NCT05827016) is now testing iberdomide as a maintenance therapy to prevent disease progression after stem cell transplant in newly diagnosed patients. The randomized study will compare iberdomide to Revlimid (lenalidomide).
Iberdomide side effects
Across various patient groups and treatment combinations, low blood cell counts have been among the most common side effects observed in people given iberdomide.
In newly diagnosed patients who received iberdomide with dexamethasone and Darzalex, other common side effects included:
- infections
- swelling
- constipation
- rash
- fatigue
- fever
- nausea
- diarrhea
- back pain
- insomnia
These side effects were similar to those observed in RRMM patients given iberdomide with dexamethasone.
Rare Cancer News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by