Etentamig for multiple myeloma
What is etentamig for multiple myeloma?
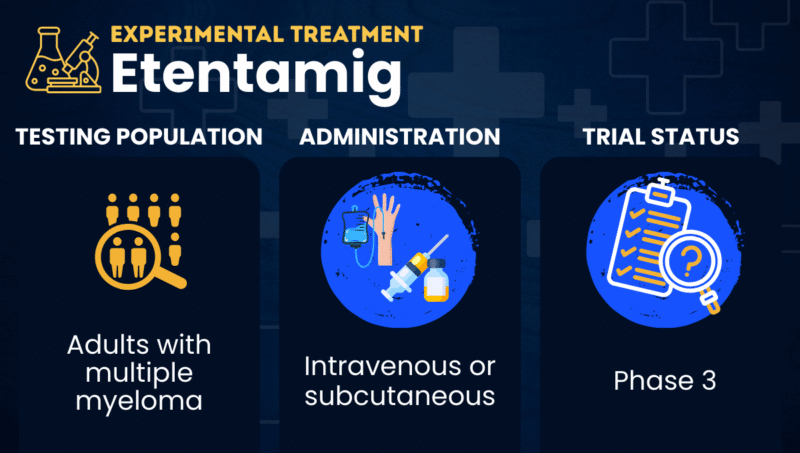
Etentamig is an experimental antibody-based therapy being tested for multiple myeloma in people who are newly diagnosed or in those whose disease has returned (relapsed) or failed to respond to prior treatment (refractory).
Multiple myeloma is a blood cancer in which plasma cells, which normally produce antibodies that help the body fight infections, grow uncontrollably and become cancerous. While immune T-cells can normally kill abnormal cells, myeloma cells are particularly adept at evading the immune system.
Etentamig is a bispecific antibody designed to help T-cells recognize and destroy the cancerous cells. It does so by simultaneously binding the CD3 protein on T-cells and BCMA on plasma cells, bringing the two cell types into contact. This triggers T-cells to release toxic proteins that kill cancer cells and produce signals that boost the immune response.
This next-generation bispecific antibody is designed to help reduce the risk of cytokine release syndrome (CRS), an immune reaction that can sometimes happen with immunotherapies and be serious. It also stays active in the body longer, meaning patients may require fewer doses than with older immunotherapy options.
Abbvie is developing etentamig for use on its own or in combination with other therapies. Although etentamig is typically administered as an intravenous (into-the-vein) infusion in current trials, researchers are also testing a subcutaneous (under-the-skin) version that would make treatment more convenient for patients.
Therapy snapshot
| Treatment name | Etentamig |
| Administration | Intravenous infusion and subcutaneous injection |
| Clinical testing | In Phase 3 testing for relapsed or refractory multiple myeloma and in Phase 2/3 trials for newly diagnosed patients |
How will etentamig be administered in multiple myeloma?
Most studies of etentamig in multiple myeloma are testing it as an intravenous infusion. Based on earlier studies, the ongoing Phase 3 CERVINO trial is testing a 60 mg dose given every four weeks as a standalone treatment. However, doses may vary when etentamig is used in combination with other treatments.
A small Phase 1 study (NCT06223516) is also assessing whether administering etentamig via subcutaneous injection is as effective as the intravenous infusion.
Etentamig in multiple myeloma clinical trials
Several ongoing trials are assessing etentamig in various multiple myeloma clinical settings. These include:
- The Phase 1/2 M22-466 study (NCT03933735) enrolled 220 adults with relapsed or refractory multiple myeloma who had received at least three prior lines of therapy. Participants received varying doses of intravenous etentamig, and early results indicated that 60 mg administered every four weeks was the optimal dose for future studies. A pooled analysis of 146 patients who took part in M22-466 and another Phase 1 study, called M24-108 (NCT05650632), suggested that 66% of patients responded to treatment, and more than half were alive and hadn’t experienced disease progression over a median follow-up of 13 months.
- The CERVINO Phase 3 trial (NCT06158841) is building on these results and aims to confirm the effectiveness of etentamig compared with standard combination therapies. About 380 adults with relapsed or refractory multiple myeloma who have received at least two prior therapies will take part. The main goals are to determine the percentage of participants who respond to treatment in each arm and how long patients live without signs of disease progression. Top-line data are expected in 2027.
- The Phase 1b Kilimanjaro trial (NCT05259839) is testing etentamig in combination with approved therapies in 283 participants with relapsed or refractory multiple myeloma. In one arm of the trial, which is studying etentamig plus Pomalyst (pomalidomide) and dexamethasone, 81% of participants experienced at least a partial response to the treatment. Another arm tested etentamig in combination with Darzalex (daratumumab) and dexamethasone, and 71% of participants responded to treatment.
- The Phase 2/3 M25-586 study (NCT07095452) is also testing etentamig as part of a combination therapy, but in newly diagnosed patients who are ineligible for a stem cell transplant. In a Phase 2 part, participants will receive one of three doses of etentamig in combination with Darzalex Faspro (daratumumab and hyaluronidase-fihj). Then, the Phase 3 portion will compare the optimal dose of etentamig plus Darzalex Faspro against an approved Darzalex Faspro combination. The main goals are to assess safety, response rates, and progression-free survival.
Etentamig side effects
Etentamig is still being tested in clinical trials, so its safety profile in people with multiple myeloma is not fully established.
So far, the most common side effects, some of them serious, reported in early trials have been related to blood cell counts. These included:
- low levels of neutrophils and lymphocytes, two types of white blood cells
- low levels of red blood cells
- low platelet counts
- infections, including pneumonia and sepsis
About 30%-43% of participants experienced CRS. While this reaction can be serious or life-threatening, all cases so far have been mild or moderate. A few cases (5%-7%) of a neurological complication called immune effector cell-associated neurotoxicity syndrome were also reported, but most were mild or moderate in severity.
Rare Cancer News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by