Empliciti (elotuzumab) for multiple myeloma
What is Empliciti for multiple myeloma?
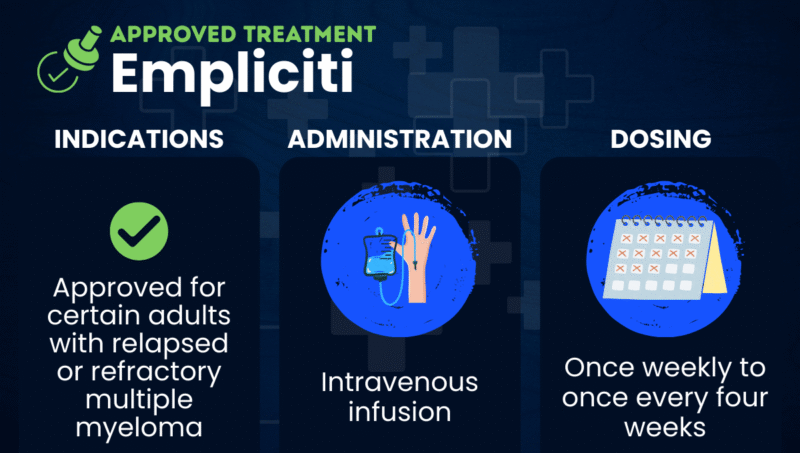
Empliciti (elotuzumab) is an infusion therapy approved for use in combination regimens for adults with multiple myeloma that did not respond to (refractory) or returned after (relapsed) other treatments.
The antibody-based therapy targets the signaling lymphocytic activation molecule family member 7 (SLAMF7), a protein found at high levels on the surface of cancerous myeloma cells and certain immune cells, including natural killer (NK) cells.
By binding to SLAMF7, Empliciti simultaneously activates NK cells and marks myeloma cells for recognition and destruction by NK cells and other immune cells that produce SLAMF7. This double-action approach is expected to help slow down or control the cancer.
Administered via into-the-vein (intravenous) infusions, the therapy is marketed by Bristol Myers Squibb.
Therapy snapshot
| Brand name | Empliciti |
| Chemical name | Elotuzumab |
| Usage | Used to treat certain adults with refractory or relapsed multiple myeloma |
| Administration | Intravenous infusion |
Who can take Empliciti?
Empliciti is approved in the U.S. as part of combination regimens to treat adults with relapsed or refractory multiple myeloma (RRMM). The therapy is used in combination with:
- Revlimid (lenalidomide) and dexamethasone for patients who have received one to three prior lines of therapy
- Pomalyst (pomalidomide) and dexamethasone for those who were previously given two or more lines of therapies, including Revlimid and a proteasome inhibitor (PI)
Empliciti’s U.S. label does not mention any contraindications. However, because the therapy is always used along with Revlimid or Pomalyst, which can cause birth defects, strict pregnancy‑prevention rules apply. In the U.S., these partner therapies are available only through restricted distribution programs.
How is Empliciti administered?
Empliciti is administered as an intravenous infusion by a healthcare provider in repeating 28‑day cycles until the disease progresses or there is unacceptable toxicity. The precise dosing regimen depends on the usage:
- when used in combination with Revlimid and dexamethasone, Empliciti is given at a dose of 10 mg/kg once a week for the first two cycles, and then every two weeks in subsequent cycles
- when used in combination with Pomalyst and dexamethasone, the first two cycles of Empliciti are the same, but subsequent cycles involve the administration of a 20 mg/kg dose every four weeks
Before each Empliciti infusion, patients receive medicines such as dexamethasone, antihistamines, and acetaminophen to lower the risk of infusion reactions, and they are monitored during and after the infusion.
Empliciti in clinical trials
Data from two global clinical trials supported Empliciti’s approvals for adults with RRMM. Both showed that adding Empliciti to standard combination regimens helped more people respond to treatment and live longer without their myeloma getting worse compared with the same regimens that didn’t include Empliciti.
- The Phase 3 ELOQUENT‑2 study (NCT01239797) demonstrated that Empliciti plus Revlimid and dexamethasone was significantly superior to Revlimid and dexamethasone alone at lowering the risk of disease progression or death (by 30%) and improving response rates (79% vs. 66%) in adults with RRMM who had received one to three previous lines of therapy. Later analyses showed that the Empliciti combo was linked to an 18% lower risk of death. These benefits were sustained for at least four years after starting treatment.
- The Phase 2 ELOQUENT‑3 study (NCT02654132) showed that adding Empliciti to Pomalyst and dexamethasone significantly reduced the risk of disease progression or death by 46% and doubled response rates (53% vs. 26%) relative to Pomalyst and dexamethasone alone in adults given at least two prior therapy lines, including Revlimid and a PI. Further analyses indicated that adding Empliciti to this treatment regimen significantly reduced the risk of death by 41%.
Empliciti side effects
The most common side effects of Empliciti in people with multiple myeloma vary depending on the regimen in which it is used.
When used in combination with Revlimid and dexamethasone, common side effects include:
- fatigue
- diarrhea
- constipation
- fever
- peripheral neuropathy, or nerve damage that can cause abnormal sensations, such as tingling or numbness
- cough
- common cold and other upper respiratory infections
- reduced appetite
- pneumonia, a lung infection
When used alongside Pomalyst and dexamethasone, the most common side effects include:
- constipation
- high blood sugar levels (hyperglycemia)
The therapy’s label also comes with warnings that it could cause other potentially serious side effects, such as:
- infusion reactions
- serious infections
- non-myeloma cancers
- liver toxicity
Empliciti can also interfere with certain blood tests used to look for a complete response in certain myeloma patients.
Doctors should monitor patients regularly during treatment to catch and manage side effects early. In some cases, treatment may need to be interrupted or permanently discontinued.
Rare Cancer News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by 


