Darzalex (daratumumab) for multiple myeloma
What is Darzalex for multiple myeloma?
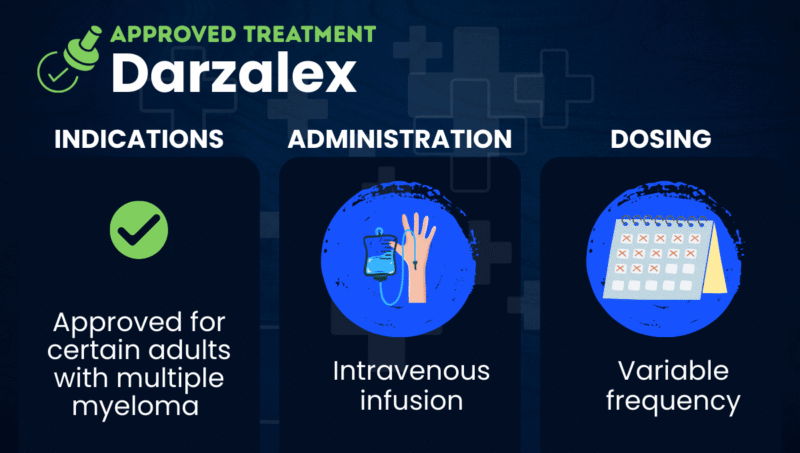
Darzalex (daratumumab) is an antibody-based therapy approved as part of several treatment regimens for adults with multiple myeloma, a type of blood cancer. It can be used in people who are newly diagnosed as well as those with relapsed or refractory multiple myeloma (RRMM), meaning the cancer has returned or has not responded to prior treatment.
Daratumumab, the treatment’s main ingredient, is an antibody that binds to CD38, a protein found at high levels on myeloma cells. This helps the immune system recognize and destroy cancer cells.
Darzalex is given as an intravenous (into-the-vein) infusion. Its developer, Johnson & Johnson, also markets a version called Darzalex Faspro, which is given as an under-the-skin injection. Darzalex Faspro can be used for people with multiple myeloma and smoldering multiple myeloma (SMM), an early form of the disease that does not yet cause symptoms.
Therapy snapshot
| Brand name | Darzalex |
| Chemical name | Daratumumab |
| Usage | Used to treat certain adults with multiple myeloma |
| Administration | Intravenous infusion |
Who can take Darzalex?
Darzalex is approved in the U.S. for certain adults with multiple myeloma.
In newly diagnosed patients, Darzalex is approved to be used:
- with Revlimid (lenalidomide) and dexamethasone or Velcade (bortezomib), melphalan, and prednisone in people who are not eligible for an autologous stem cell transplant
- with Velcade, thalidomide, and dexamethasone in people who are transplant-eligible
In adults with RRMM, Darzalex is approved to be used:
- with Revlimid and dexamethasone or Velcade and dexamethasone in people who have received at least one prior therapy
- with Kyprolis (carfilzomib) and dexamethasone in people who have received one to three prior lines of therapy
- with Pomalyst (pomalidomide) and dexamethasone in people who have received at least two prior therapies, including Revlimid and a proteasome inhibitor (PI)
- as a single therapy in people who have received at least three prior lines of treatment, including a PI and an immunomodulatory agent (IMiD), or who are double-refractory to a PI and IMiD
Darzalex is contraindicated for people with a history of severe immune reactions (hypersensitivity) to daratumumab or any of its ingredients.
The use of Darzalex in combination with Revlimid, Pomalyst, or thalidomide is contraindicated during pregnancy due to the risk of fetal harm or death.
How is Darzalex administered?
Darzalex is administered by a healthcare provider as an intravenous (into-the-vein) infusion.
The recommended dose is 16 mg/kg of body weight. The first infusion typically lasts about seven hours, with later infusions lasting about three to four hours. In some cases, the first dose may be split into two 8 mg/kg doses given on consecutive days, each taking a little more than four hours.
Infusion frequency varies by treatment plan. Generally, Darzalex is given weekly at first, then every two to four weeks. Total treatment duration varies, but the medication may be given as follows:
- For RRMM and newly diagnosed, transplant-ineligible myeloma: until disease progression occurs
- In newly diagnosed, transplant-eligible myeloma: for a set number of doses before and after stem cell transplant
Patients also receive medications before each infusion to help reduce the risk of side effects and may be instructed to take oral steroid medications for a few days afterward.
Antiviral medications to prevent reactivation of herpes zoster — the cause of shingles — should be initiated when starting Darzalex and continued until three months after treatment ends.
Darzalex in clinical trials
Data from several clinical trials helped support the approval of Darzalex for treating multiple myeloma in different patient groups.
Studies involving people newly diagnosed with myeloma included:
- The Phase 3 MAIA trial (NCT02252172) showed that adding Darzalex to Revlimid and dexamethasone reduced the risk of disease progression or death by 44% in transplant-ineligible patients.
- The Phase 3 ALCYONE trial (NCT02195479) found similar benefits in this patient population when Darzalex was combined with Velcade, melphalan, and prednisone.
- The Phase 3 CASSIOPEIA trial (NCT02541383) showed that adding Darzalex to Velcade, thalidomide, and dexamethasone led to higher rates of complete treatment response and reduced the risk of disease progression or death in patients eligible for a stem cell transplant, compared with the same regimen without Darzalex.
Studies involving people with RRMM included:
- The Phase 3 POLLUX trial (NCT02076009) showed that Darzalex plus Revlimid and dexamethasone significantly extended survival without disease progression in people who received at least one prior therapy.
- The Phase 3 CASTOR trial (NCT02136134) found similar benefits when Darzalex was combined with Velcade and dexamethasone.
- The Phase 3 CANDOR trial (NCT03158688) showed similar benefits when Darzalex was combined with Kyprolis and dexamethasone for people who had received one to three prior lines of therapy.
- The Phase 1 EQUULEUS study (NCT01998971) tested multiple treatment regimens, including the combination of Darzalex, Pomalyst, and dexamethasone in people who had received at least two prior lines of treatment. About 60% of patients experienced at least a partial response.
- The Phase 2 SIRIUS trial (NCT01985126) tested Darzalex as a monotherapy in people who had received at least three prior lines of treatment. About 30% had at least a partial response to treatment.
- The Phase 2 GEN501 study (NCT00574288) showed Darzalex monotherapy led to a 36% response rate in people who had received at least two prior lines of treatment.
Darzalex side effects
The most common side effects associated with Darzalex include:
- upper respiratory infections
- low blood cell counts
- infusion-related reactions
- diarrhea or constipation
- nerve-related symptoms, such as numbness, tingling, or burning sensations
- fatigue
- swelling
- nausea
- cough
- fever
- shortness of breath
- weakness or lack of energy
Darzalex also comes with a warning for potentially serious side effects, including:
- infusion-related reactions, some of which could be life-threatening
- low counts of neutrophils, a type of infection-fighting immune cell, which may increase the risk of serious or fatal infections
- low platelet counts, which may increase the risk of bleeding
- harm to a developing fetus if used during pregnancy
Patients will be monitored for these complications, and treatment may be delayed, slowed, or stopped if serious side effects occur.
Females of reproductive potential should use effective contraception during treatment and for three months after the final dose of Darzalex.
Rare Cancer News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by