Iopofosine I 131 for multiple myeloma
What is iopofosine I 131 for multiple myeloma?
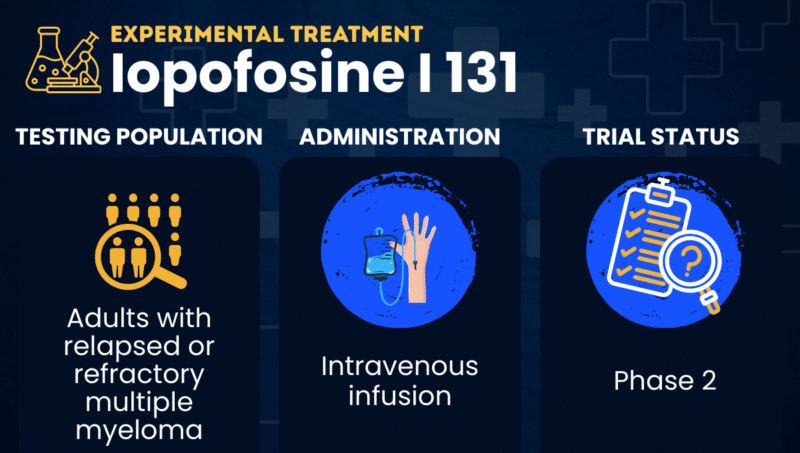
Iopofosine I 131 is an experimental targeted radiotherapy being developed for people with relapsed or refractory multiple myeloma, meaning their cancer has returned or has not responded to prior treatments.
Given by intravenous infusion, or into the bloodstream, iopofosine I 131 is a phospholipid drug conjugate with two key components: a fatty molecule called a phospholipid ether attached to the radioactive compound iodine-131 (I-131), which emits radiation capable of killing cells.
The therapy, formerly known as CLR 131, is designed to exploit differences between myeloma cells and healthy cells. In particular, myeloma cells contain specialized regions in their outer membrane known as lipid rafts. These structures are involved in cell signaling, survival, drug resistance, and communication with the surrounding environment, helping cells thrive.
The phospholipid ether in iopofosine I 131 is designed to preferentially bind these lipid rafts, allowing the compound to enter myeloma cells. Once inside the cell, the radioactive iodine delivers localized radiation intended to damage and kill the cancer cell while minimizing exposure to healthy tissue.
Iopofosine I 131 received orphan drug and fast-track designations in the U.S. and has orphan drug status in the European Union. These regulatory programs aim to speed the development and review of therapies for serious conditions with unmet medical needs.
The therapy is being developed by Cellectar Biosciences and is also being investigated for other blood cancers and pediatric glioma, a type of brain cancer.
Therapy snapshot
| Treatment name | Iopofosine I 131 |
| Administration | Intravenous infusion |
| Clinical testing | In Phase 2 testing for relapsed or refractory multiple myeloma |
How will iopofosine I 131 be administered in multiple myeloma?
In clinical trials involving people with relapsed or refractory multiple myeloma, iopofosine I 131 has been given as an intravenous infusion, typically lasting about 15-30 minutes.
Researchers have evaluated different dosing strategies, including a single larger dose and fractionated dosing, in which the total radiation dose is split into two infusions.
Because the thyroid gland naturally absorbs iodine, participants have received medications to reduce the risk of thyroid damage from radioactive iodine. Low doses of the corticosteroid dexamethasone have also been administered in some study groups.
Iopofosine I 131 in multiple myeloma clinical trials
Two main clinical studies have evaluated iopofosine I 131 in people with relapsed or refractory multiple myeloma.
A Phase 1 dose-escalation study evaluated different dosing regimens to establish optimal dosing strategies for future studies, and an ongoing Phase 2 clinical trial is testing iopofosine I 131 across several blood cancers, including myeloma. Both studies have enrolled heavily pretreated multiple myeloma patients.
- The early Phase 1 study (NCT02278315) enrolled 31 adults who had received a median of five prior lines of therapy. The study tested increasing dose levels, using a single infusion in the first four dose groups, and later switching to fractionated dosing to allow higher total radiation exposure while managing side effects. Data showed that patients given a single infusion at any of the tested doses lived for a median of 22 months. However, fractionated dosing enabled patients to receive higher radiation doses with fewer severe side effects. In the group given a high fractionated dose of 37.5 millicurie (mCi) per square meter of body surface, all had at least stable disease, and 50% experienced a partial response to treatment.
- The open-label Phase 2 CLOVER-1 study is evaluating iopofosine I 131 in several B-cell cancers, including people with relapsed or refractory multiple myeloma. These patients had received at least five previous lines of therapy and had failed to respond to at least three classes of treatment. Results showed that among patients who received a total dose of 60 mCi or greater, 96% had at least stable disease, and 32% responded to treatment. A total of nine participants were also refractory to a fourth treatment class, and one-third of them responded to treatment.
Iopofosine I 131 side effects
Iopofosine I 131 is still being tested in clinical trials for multiple myeloma, so its side-effect profile is not fully established.
To date, safety data are available for 64 patients with relapsed or refractory multiple myeloma. The most common side effects were low levels of certain blood components, including platelets, red blood cells, neutrophils, and white blood cells. Some of these were serious.
Other side effects reported, most of them mild to moderate, included:
- diarrhea
- fatigue
- low calcium or phosphate levels
- shortness of breath
- headache
Rare Cancer News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by