Carvykti (ciltacabtagene autoleucel) for multiple myeloma
What is Carvykti for multiple myeloma?
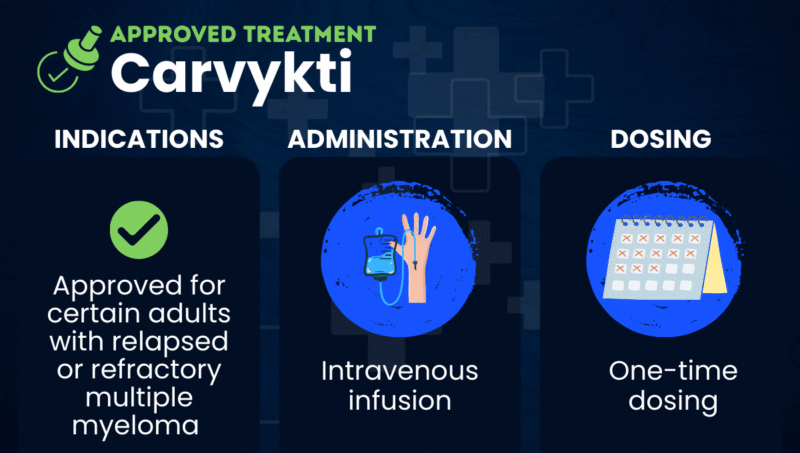
Carvykti (ciltacabtagene autoleucel) is an approved CAR T-cell therapy that’s used to treat certain adults with multiple myeloma that has returned after (relapsed) or not responded to (refractory) other treatments.
Myeloma is a type of blood cancer that arises when immune plasma cells grow out of control in the bone marrow.
As a CAR T-cell therapy, Carvykti is designed to help the body’s own immune system more effectively attack and kill these cancer cells. It involves collecting a patient’s T-cells — a type of immune cell with innate cancer-killing abilities — and engineering them in the lab with a special chimeric antigen receptor, or CAR, that helps them more specifically target and destroy myeloma cells. The CAR used in Carvykti is specifically designed to bind to the B-cell maturation antigen (BCMA) protein, which is found at high levels on the surface of myeloma cells.
When returned to the patient via a one-time into-the-vein (intravenous) infusion, the engineered T-cells then find the cancerous cells containing BCMA and cause their destruction.
Legend Biotech and Johnson & Johnson Innovative Medicine jointly developed Carvykti. The companies are currently investigating Carvykti’s potential use in other myeloma patients, including those newly diagnosed with the blood cancer.
Therapy snapshot
| Brand name | Carvykti |
| Chemical name | Ciltacabtagene autoleucel |
| Usage | Used to treat certain adults with relapsed or refractory multiple myeloma |
| Administration | Intravenous infusion |
Who can take Carvykti?
Carvykti is approved in the U.S. for the treatment of adults with relapsed or refractory multiple myeloma who have received at least one prior line of therapy, including a proteasome inhibitor and an immunomodulatory agent, and who failed to respond to Revlimid (lenalidomide).
The therapy is similarly approved in the European Union and Canada, although specific indications may vary somewhat.
There are no specific contraindications for Carvykti’s use. However, the therapy comes with a boxed warning that it may cause a number of serious complications that could be life-threatening, including severe inflammatory and immune reactions, neurological problems, secondary blood cancers, and low blood cell counts.
How is Carvykti administered?
Carvykti is always administered at a treatment center with the appropriate equipment and expertise to manage the infusion and any complications that may arise. Carvykti’s recommended dose range is 0.5 to 1 million CAR T-cells per kilogram of body weight per infusion.
Treatment with Carvykti is a five-step process that takes about two to three months to complete:
- Cell collection: T-cells are collected from the patient’s blood over a period of three to six hours.
- Cell modification: The cells are engineered in the lab over the next four to five weeks. A doctor may prescribe other myeloma treatments during that time.
- Pre-infusion treatment: A few days before receiving Carvykti, patients receive chemotherapy, including cyclophosphamide and fludarabine, to prepare the body for treatment and make room for the new modified cells. This will be done daily for three days.
- Carvykti infusion: Two to four days after chemotherapy, Carvykti is given as a single intravenous infusion that takes 30 minutes to an hour. Before the infusion, patients may receive medications to prevent immune reactions.
- Monitoring: Patients are monitored for complications daily for a week after the infusion. They are also advised to stay near their treatment center and avoid driving for at least two weeks.
Carvykti in clinical trials
Carvykti’s approvals for multiple myeloma were mainly supported by data from two clinical trials:
- The Phase 3 CARTITUDE-4 trial (NCT04181827) evaluated Carvykti versus standard myeloma treatment regimens in 419 adults who had received one to three prior lines of therapy and had Revlimid-refractory disease. Carvykti lowered the risk of disease progression or death compared with standard care and led to higher overall treatment response rates. Other analyses showed that Carvykti was associated with a lower risk of death than standard treatments after a median of nearly three years.
- The Phase 1/2 CARTITUDE-1 trial (NCT03548207) tested the effects of Carvykti in 97 adults who had previously received at least three prior lines of therapy. The data showed that nearly all participants had at least a partial response to treatment. About one-third of patients remained alive and without cancer progression for at least five years after the treatment.
Various ongoing Phase 2 and Phase 3 clinical trials are testing Carvykti in other myeloma groups, including as an earlier-line treatment for newly diagnosed patients.
Carvykti side effects
The most common side effects associated with Carvykti include:
- fever
- cytokine release syndrome
- low levels of antibodies in the blood, which increases infection risk
- low blood pressure
- musculoskeletal pain
- fatigue
- infections, including upper respiratory infections and viral infections
- cough
- chills
- diarrhea or constipation
- nausea, vomiting, and appetite loss
- neurological problems
- headache
- fast heart rate
- dizziness
- shortness of breath
- swelling
- blood-clotting problems
Low blood cell counts are among the most common laboratory abnormalities.
All of the complications listed in the boxed warning for Carvykti may be life-threatening, so patients will be carefully monitored for them. These include:
- cytokine release syndrome (CRS), a type of systemic inflammatory response
- neurological complications, including immune effector cell-associated neurotoxicity syndrome (ICANS), Parkinsonism, or Guillain-Barré syndrome
- hemophagocytic lymphohistiocytosis and macrophage activation syndrome, which can cause organ failure from uncontrolled immune activity
- immune effector cell-associated enterocolitis, or immune-mediated damage to the gastrointestinal tract
- prolonged or recurrent low blood cell counts, which can cause bleeding and infections
- secondary blood cancers
Carvykti shouldn’t be given to people with active infections or inflammatory disorders, as this can increase the likelihood of CRS. Certain immunosuppressant treatments and supportive care may be used to manage CRS or ICANS. Low blood cell counts may require blood transfusions, medications, or a stem cell transplant.
Carvykti also comes with warnings that it may be associated with:
- hypersensitivity, or allergic-type reactions, including serious systemic reactions (anaphylaxis)
- other neurological problems
- severe, life-threatening, or fatal infections
- an increased risk of early death, with most deaths in clinical trials being related to infections
Patients will be monitored for these complications and treated as appropriate. Patients should follow their healthcare providers’ guidelines on the appropriate vaccination schedule before and after receiving Carvykti.
Carvykti is generally not recommended during pregnancy. Patients should discuss with their care providers the need for contraception after receiving Carvykti and chemotherapy.
Rare Cancer News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by