Arlocabtagene autoleucel for multiple myeloma
What is arlo-cel for multiple myeloma?
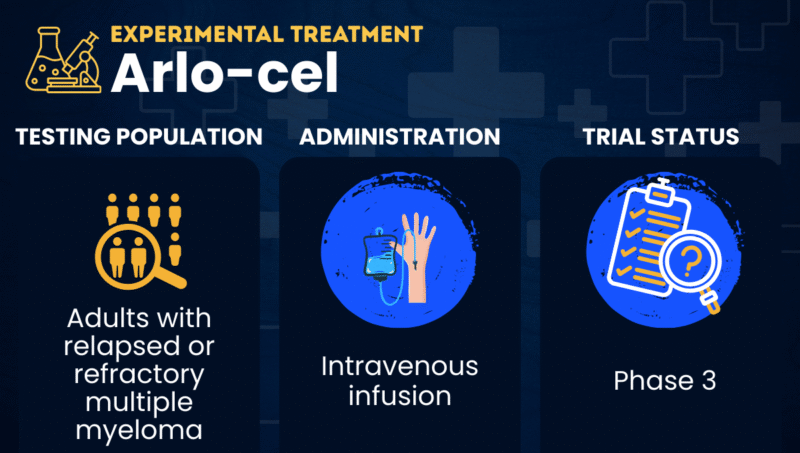
Arlocabtagene autoleucel, or arlo-cel (BMS-986393), is a CAR T-cell therapy in development for use in multiple myeloma. The treatment candidate is specifically being tested in adults with relapsed or refractory multiple myeloma (RRMM), in whom the cancer does not respond to or returns after treatment.
In multiple myeloma, immune plasma cells become cancerous and start growing out of control in the bone marrow, the spongy interior of some bones. T-cells, a type of immune cell that can target and kill cancer cells, are typically unable to effectively slow or stop this abnormal growth.
CAR T-cell therapies like arlo-cel are individualized treatments designed to help T-cells more effectively recognize and destroy cancer cells. Arlo-cel is made from a patient’s own T-cells, engineered in a lab to carry a chimeric antigen receptor, or CAR. This specialized CAR can recognize and bind to a protein called G protein-coupled receptor class C group 5 member D (GPRC5D), which is found primarily on malignant plasma cells.
When returned to the patient via a one-time intravenous, or into-the-vein, infusion, the engineered T-cells can then find and kill the cancerous plasma cells containing GPRC5D.
Bristol Myers Squibb took over the development of arlo-cel after acquiring Celgene and its subsidiary Juno Therapeutics.
Therapy snapshot
| Treatment name | Arlocabtagene autoleucel |
| Administration | Intravenous infusion |
| Clinical testing | Currently in Phase 3 testing |
How will arlo-cel be administered in multiple myeloma?
Arlo-cel is designed to be administered via a single intravenous infusion after a short round of chemotherapy that’s given to wipe out existing immune cells and prepare the body to receive the engineered T-cells.
In Phase 2 and Phase 3 clinical trials testing arlo-cel in people with RRMM, a recommended dose containing approximately 150 million CAR T-cells is being used. However, it’s still unclear if this will be the selected dose if the therapy is ultimately approved.
Arlo-cel in multiple myeloma clinical trials
The Phase 1 CC-95266-MM-001 clinical trial (NCT04674813) assessed the safety and early effectiveness of arlo-cel in adults with RRMM. Most participants had received at least three prior treatments. The first part of the trial tested several doses of arlo-cel. Based on safety and efficacy results, investigators selected a dosage of about 150 million CAR T-cells for further study.
Key results were positive:
- In 79 participants who received arlo-cel at any dose across the entire study, 87% experienced at least a partial response to treatment.
- The response rates were similar across distinct patient subgroups with different prior medication histories and disease characteristics.
- An analysis of laboratory markers suggested that arlo-cel may target cancerous plasma cells with more specificity than Abecma (idecabtagene vicleucel), an approved CAR T-cell therapy for RRMM that uses a CAR that targets a different protein on plasma cells.
Several ongoing trials are testing arlo-cel in adults with RRMM:
- The Phase 2 QUINTESSENTIAL study (NCT06297226), launched in 2024, involves about 230 individuals who previously received at least three lines of treatment, including four specific classes of medications. All participants receive an arlo-cel infusion and are followed for as long as 15 years. Investigators are assessing the percentage of participants who experience at least a partial response, with a particular focus on patients who have previously received at least four lines of treatment.
- The Phase 3 QUINTESSENTIAL-2 study (NCT06615479), begun in 2025, involves participants who received one to three prior lines of treatment. About 440 individuals are randomly receiving arlo-cel or standard of care therapies. One of the study’s main goals is assessing differences between the treatment groups in progression-free survival, or the time that participants live without experiencing disease progression.
- A Phase 1 study called CA088-1005 (NCT06121843) is testing arlo-cel in combination with other experimental therapies. The first part of the trial involves participants who have received at least three prior lines of treatment and will focus on assessing different dosing regimens and treatment combinations. Part two will then test selected, optimal doses for each combination in larger groups of participants who have previously received one to three prior treatments.
Arlo-cel side effects
Preliminary Phase 1 trial results found that, for participants who received at least half the recommended dosage of arlo-cel, the most common side effects were:
- altered blood cell counts, including low levels of red blood cells (anemia), platelets (thrombocytopenia), and a type of white blood cell called neutrophils (neutropenia)
- low potassium or phosphate levels
- diarrhea
- fatigue
- altered taste, such as metallic or bitter taste
- headache
- nausea
- changes in the skin, nails, or mouth
- cytokine release syndrome (CRS), a type of whole-body immune reaction
Low blood cell counts may make the body more vulnerable to infections, including serious infections.
Although CRS was common in this treatment group, all events were mild or moderate.
A minority of participants (about 10%) experienced neurological complications, such as a condition called immune effector cell-associated neurotoxicity syndrome, or ICANS.
Rare Cancer News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by